The successful completion of many Pharmacopeial tests and assays requires adjustment to or maintenance of a specified pH by the addition of buffer solutions. In pH measurements, standard buffer solutions are required for reference purposes. For convenience, the preparation of these solutions is in some instances described in the sections in which their use is specified; i.e., five separate phosphate buffers are described under
Antibiotics—Microbial Assays  81
81
, and several miscellaneous single-purpose solutions are described in the individual monographs.
A solution is said to be buffered if it resists changes in the activity of an ion on the addition of substances that are expected to change the activity of that ion. Buffers are substances or combinations of substances that impart this resistance to a solution. Buffered solutions are systems in which the ion is in equilibrium with substances capable of removing or releasing the ion.
Buffer capacity refers to the amount of material that may be added to a solution without causing a significant change in ion activity. It is defined as the ratio of acid or base added (in gram-equivalents per liter) to the change in pH (in pH units). The capacity of a buffered solution is adjusted to the conditions of use, usually by adjustment of the concentrations of buffer substances.
Buffers are used to establish and maintain an ion activity within narrow limits. The most common systems are used (a) to establish hydrogen-ion activity for the calibration of pH meters, (b) in the preparation of dosage forms that approach isotonicity, (c) in analytical procedures, and (d) to maintain stability of various dosage forms. Buffers used in physiological systems are carefully chosen so as not to interfere with pharmacological activity of the medicament or normal function of the organism. It is essential that buffers used in chemical analysis be compatible with the substance determined and the reagents used.
 Standard Buffer Solutions
Standard Buffer Solutions
—Standard solutions of definite pH are readily available in buffer solutions prepared from the appropriate reagents. In addition, buffer solutions, buffer tablets, and buffer solids may be obtained from commercial sources in convenient prepackaged form. Such preparations are available for the entire working range in pharmaceutical analysis, but are not recommended for pH meter standardization (see
pH  791
791
).
The required reagents are described in the section,
Reagents. Previously dry the crystalline reagents, except the boric acid, at 110
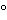
to 120
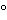
for 1 hour.
NOTE—Where water is specified for solution or dilution of test substances in pH determinations, use carbon dioxide-free water.
Store the prepared solutions in chemically resistant, tight containers such as Type I glass bottles. Use the solutions within 3 months.
Standard Buffer Solutions for various ranges between pH 1.2 and 10.0 may be prepared by appropriate combinations of the solutions described herein, used in the proportions shown in the accompanying table. The volumes shown in the table are for 200 mL of buffer solution, except that the volumes shown for Acetate Buffer are used to prepare 1000 mL of buffer solution.
-
Hydrochloric Acid, 0.2 M, and Sodium Hydroxide, 0.2 M—Prepare and standardize as directed under Volumetric Solutions.
-
Potassium Biphthalate, 0.2 M—Dissolve 40.85 g of potassium biphthalate [KHC6H4(COO)2] in water, and dilute with water to 1000 mL.
-
Potassium Phosphate, Monobasic 0.2 M—Dissolve 27.22 g of monobasic potassium phosphate (KH2PO4) in water, and dilute with water to 1000 mL.
-
Boric Acid and Potassium Chloride, 0.2 M—Dissolve 12.37 g of boric acid (H3BO3) and 14.91 g of potassium chloride (KCl) in water, and dilute with water to 1000 mL.
-
Potassium Chloride, 0.2 M—Dissolve 14.91 g of potassium chloride (KCl) in water, and dilute with water to 1000 mL.
-
Acetic Acid, 2 N—Prepare and standardize as directed under Volumetric Solutions.

