á16ñAUTOMATED METHODS OF ANALYSIS
Where a sufficiently large number of similar units are to be subjected routinely to the same type of examination,automated methods of analysis may be far more efficient and precise than manual methods.Such automated methods have been found especially useful in testing the content uniformity of tablets and capsules and in facilitating methods requiring precisely controlled experimental conditions.Many manufacturing establishments,as well as the laboratories of regulatory agencies,have found it convenient to utilize automated methods as alternatives to Pharmacopeial methods (see Proceduresunder Tests and Assaysin the General Notices and Requirements).In addition,the detection system and calculation of results for automated methods are often computerized.
Before an automated method for testing an article is adopted as an alternative,it is advisable to ascertain that the results obtained by the automated method are equivalent in accuracy and precision to those obtained by the prescribed Pharmacopeial method,bearing in mind the further principle stated in the General Notices and Requirementsthat “where a difference appears,or in the event of dispute,only the result obtained by the procedure given in this Pharmacopeia is conclusive.”
It is necessary to monitor the performance of the automated analytical system continually by assaying standard preparations of known composition frequently interspersed among the test preparations.Where immiscible solvents are employed in the automated apparatus for rapid extractions,they are often separated for analysis before complete extraction is attained,and the chemical reactions utilized in automated methods rarely are stoichiometric.Both the accuracy and the precision of the determinations depend upon precise adjustment of the equipment,so maintained that all standard and test preparations are exposed to identical physical and chemical manipulations for identical time intervals.Excessive variability in the response of the standard preparations indicates that the analytical system is malfunctioning and that the test results are therefore invalid.However,where automated systems are shown to operate reliably,the precision of the automated method may surpass that of the manual procedure employing the same basic chemistry.
Many of the manual methods given in this Pharmacopeia can be adapted for use in automated equipment incorporating either discrete analyzers or continuous flow systems and operating under a variety of conditions.On the other hand,an analytical scheme devised for a particular automated system may not be readily transposable for use either in a manual procedure or in other types of automated equipment.
The apparatus required for manual methods is,in general,less complicated than the apparatus of automated systems,even those systems used for the direct automated measurement of a single analyte (i.e.,the substance being determined or analyzed for)in a binary mixture.However,because of their versatility,automated systems designed for the rapid determination of a specified substance often can be readily modified by the addition of suitable modules and accessories to permit the determination of one or more additional substances in a dosage form.Such extended systems have been utilized,for example,in the automated analysis of articles containing both estrogens and progestogens.
The accompanying pertinent diagrams represent examples of automated methods.Diagrams for official methods are reproduced here rather than in the individual monographs.The descriptions of the procedural details in these methods exemplify the general approach in automated analysis applicable to dosage forms.It should be noted that the diagrams,with many minutiae,are an indispensable part of the directions for conducting the analysis.
DIAGRAMS
The diagrams shown below are arranged in alphabetic order by the name of the drug first mentioned,where the diagram is for a procedure for a specific article.Where there is no procedure in this chapter for a particular diagram,reference is to be made to the named monograph.
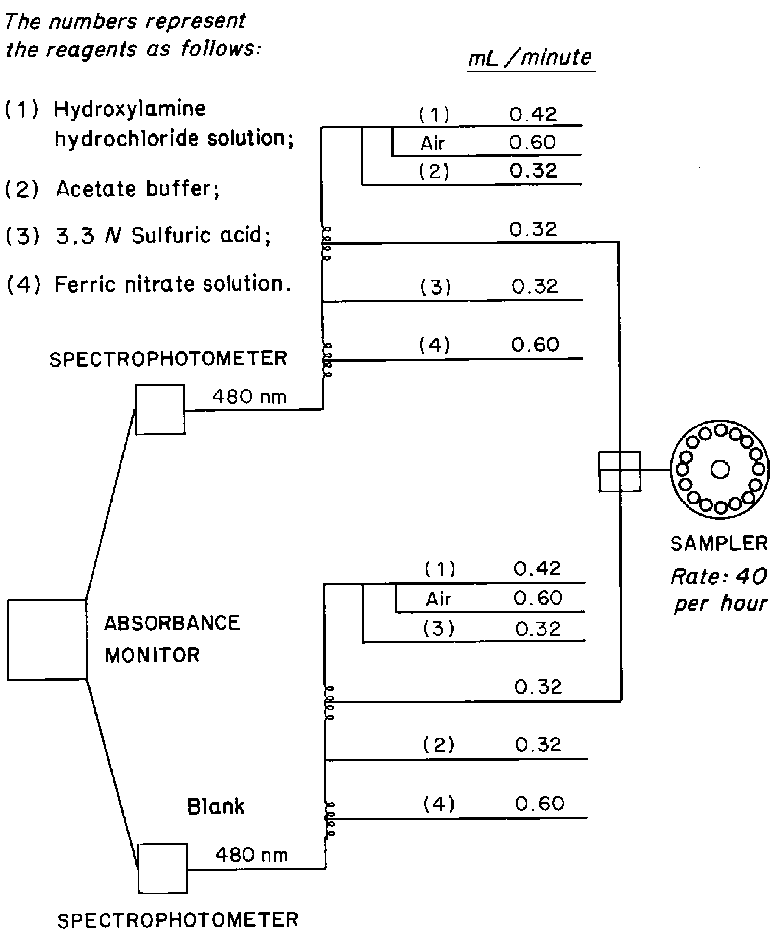
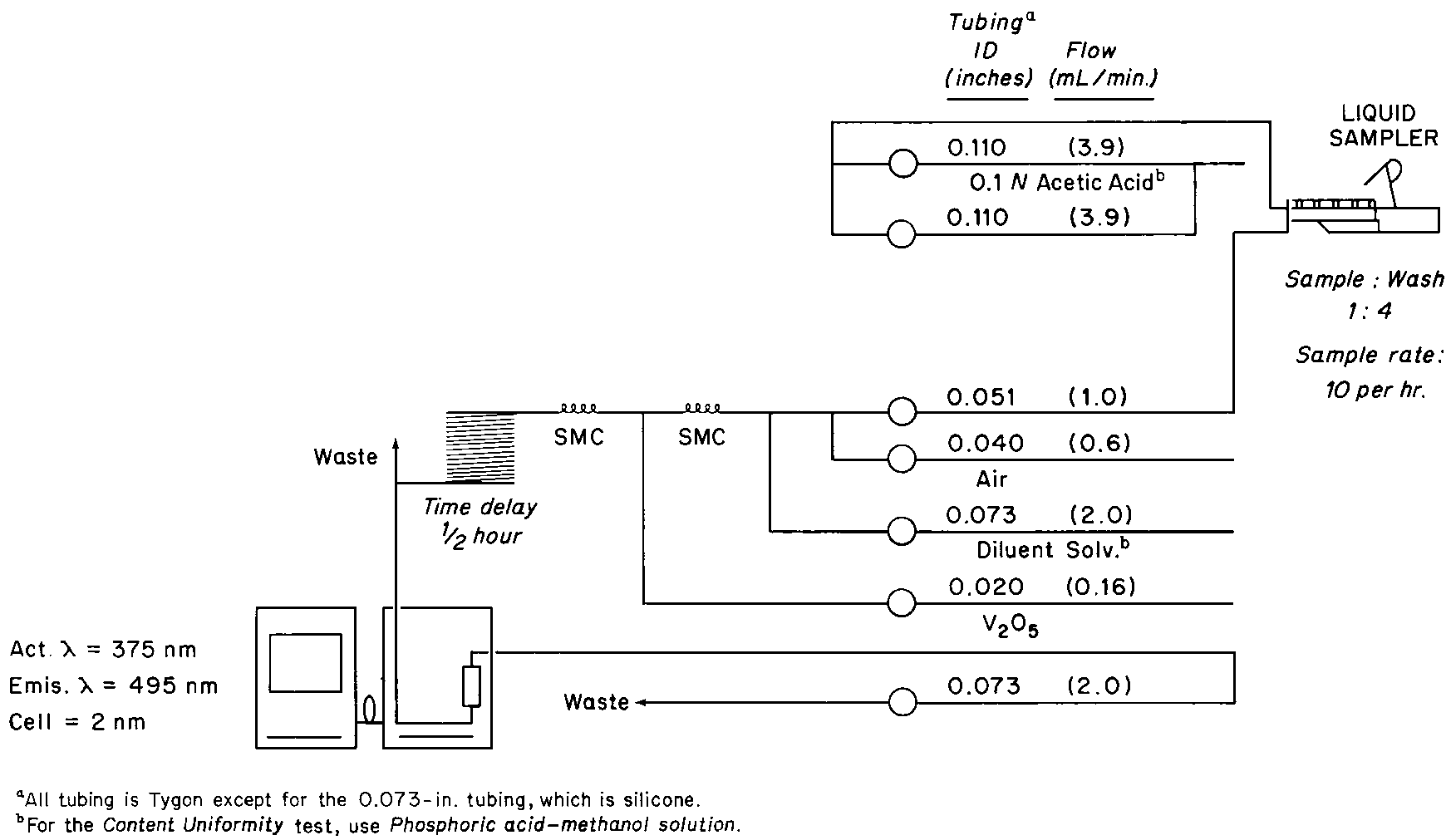
ANTIBIOTICS—HYDROXYLAMINE ASSAY
The following procedure is applicable for the assay of those Pharmacopeial antibiotics,such as cephalosporins and penicillins,that possess the beta-lactam structure.
Apparatus—
Automatic analyzer consisting of (1)a liquid sampler,(2)a proportioning pump,(3)suitable spectrophotometers equipped with matched flow cells and analysis capability at 480nm,(4)a means of recording spectrophotometric readings,and/or computer for data retrieval and calculation,and (5)a manifold consisting of the components illustrated in the accompanying pertinent diagram.
Reagents—
Hydroxylamine Hydrochloride Solution—
Dissolve 20g of hydroxylamine hydrochloride in 5mLof polyoxyethylene (23)lauryl ether solution (1in 1000),and add water to make 1000mL.
Acetate Buffer—
Dissolve 173g of sodium hydroxide and 20.6g of sodium acetate in water to make 1000mL.Dilute 75mLof this solution with water to 500mL,and mix.
Ferric Nitrate Solution—
Suspend 233g of ferric nitrate in about 600mLof water,add 2.8mLof sulfuric acid,stir until the ferric nitrate is dissolved,add 1mLof polyoxyethylene (23)lauryl ether,dilute with water to 1000mL,and mix.
USP Reference Standards á11ñ—
Use the USP Reference Standard as directed in the individual monograph.
Standard Preparation—
Unless otherwise directed in the individual monograph,dissolve an accurately weighed quantity of the USP Reference Standard in water,and quantitatively dilute with water to obtain a solution having a known concentration of about 1mg per mL.
Assay Preparation—
Unless otherwise directed in the individual monograph,using the specimen under test,prepare as directed under Standard Preparation.
Procedure—
With the sample line pumping water,the other lines pumping their respective reagents,and the spectrophotometer set at 480nm,standardize the system until a steady absorbance baseline has been established.Transfer portions of the Standard Preparationand the Assay Preparationto sampler cups,and place in the sampler.Start the sampler,and conduct determinations of the Standard Preparationand the Assay Preparationtypically at the rate of 40per hour,using a ratio of about 2:1for sample and wash time.Calculate the potency by the formula given in the individual monograph,in which Cis the concentration,in mg per mL,of USP Reference Standard in the Standard Preparation;Pis the potency,in µg per mg,of the USP Reference Standard;and AUand ASare the absorbances,corrected for the absorbances of the respective blanks,of the solutions from the Assay Preparationand the Standard Preparation,respectively.
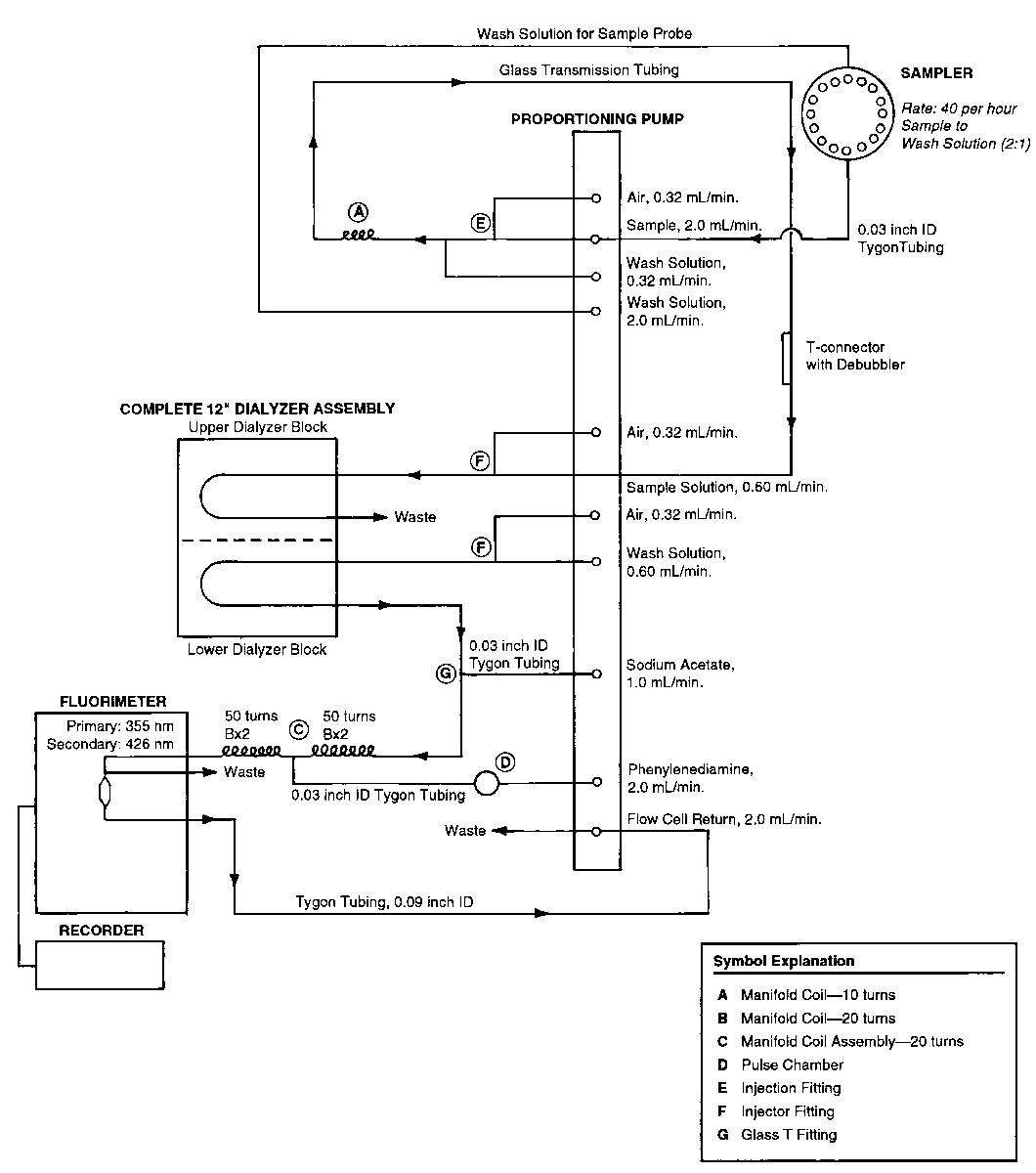
ASSAY FOR ASCORBIC ACID
The following procedure is applicable for the assay of ascorbic acid in Pharmacopeial multivitamin-minerals combination products (solid and liquid dosage forms)that contain components that interfere in other methods of assay.
Apparatus—
Automatic analyzer consisting of (1)a liquid sampler;(2)a proportioning pump;(3)a suitable fluorimeter equipped with a flow cell and filters:primary—335nm,and secondary—426nm;(4)a means of recording fluorimeter readings;and (5)a manifold consisting of the components illustrated in the accompanying pertinent diagram.
Reagents—
Extracting Solution—
Dissolve 600g of metaphosphoric acid in 1200mLof water.Add 400mLof glacial acetic acid,dilute with water to 2000mL,and mix.
Dilute Extracting Solution—
Dissolve 60g of metaphosphoric acid in 1200mLof water.Add 160mLof glacial acetic acid,dilute with water to 2000mL,and mix.
Surfactant Solution—
Prepare a 30%solution of polyoxyethylene (23)lauryl ether by melting 150g in a container on a steam bath and slowly adding approximately 250mLof water with continuous stirring.Cool and dilute with water to make 500mL.
Wash Solution—
Add 1mLof Surfactant Solutionto 3000mLof Dilute Extracting Solution,and mix.
Carbon Extraction Solution—
Dissolve 60g of metaphosphoric acid in 1200mLof water.Add 160mLof glacial acetic acid,and mix.Add 33g of activated charcoal powder,mix,and dilute with water to 2000mL.Continually mix the solution at a rate that maintains homogeneity.
Sodium Acetate Solution—
Dissolve 500g of sodium acetate trihydrate in water to make 1000mL,mix,and filter.
Phenylenediamine Solution—
Dissolve 200mg of o-phenylenediamine dihydrochloride in water to make 1000mL,and mix.Prepare fresh daily.
USP Reference Standards á11ñ—
USP Ascorbic Acid RS.
Standard Stock Solution—
Dissolve an accurately weighed quantity of USP Ascorbic Acid RSin Dilute Extracting Solutionto obtain a solution having a known concentration of about 0.1mg per mL.
Standard Preparations—
Transfer 10.0,20.0,30.0,40.0,and 50.0mLof Standard Stock Solutionto separate 100-mLvolumetric flasks,dilute the contents of each flask with Carbon Extracting Solutionto volume,mix,and filter to obtain Standard Preparations A,B,C,D,and Ehaving known concentrations of 10µg,20µg,30µg,40µg,and 50µg of USP Ascorbic Acid RSper mL,respectively.
Assay Preparation—
For Liquid Preparations—
Transfer an accurately measured volume of the liquid preparation,equivalent to 150mg of ascorbic acid,to a 100-mLvolumetric flask.Add 10mLof Extracting Solutionand 6mLof glacial acetic acid.Dilute with water to volume,and mix.Transfer 2.0mLof this solution to a 100-mLvolumetric flask,dilute with Carbon Extracting Solutionto volume,mix,and filter.
For Tablet Preparations—
Weigh and finely powder not fewer than 20Tablets.Transfer an accurately weighed quantity of the powder,equivalent to about 250mg of ascorbic acid,to a 250-mLvolumetric flask.Add 25mLof Extracting Solution,15mLof glacial acetic acid,and about 100mLof water,and swirl to mix.Heat for 15minutes in a 70 water bath,swirling after about 7minutes.Cool,and dilute with water to volume.Transfer 2.0mLof this solution to a 100-mLvolumetric flask,dilute with Carbon Extracting Solutionto volume,mix,and filter.
water bath,swirling after about 7minutes.Cool,and dilute with water to volume.Transfer 2.0mLof this solution to a 100-mLvolumetric flask,dilute with Carbon Extracting Solutionto volume,mix,and filter.
For Capsule Preparations—
Empty the contents,if necessary by cutting open with a sharp blade,of not fewer than 20Capsules in a suitable container,and mix thoroughly.Transfer a portion of the capsule contents,equivalent to about 250mg of ascorbic acid,to a 250-mLvolumetric flask,and proceed as directed for Tablets above,beginning with “Add 25mLof .”
Procedure—
With the sample line pumping the Wash Solution,the other lines pumping their respective reagents,and the fluorimeter equipped with proper filters,standardize the system by pumping until a steady baseline has been established.Transfer portions of theStandard Preparationsand the Assay Preparationto sample cups,and place in the sampler.Start the sampler,and conduct determinations of each Standard Preparationand the Assay Preparationat the rate of 40per hour,using a ratio of about 2:1for sample and wash time.Derive a standard response line by plotting the respective Standard Preparationconcentration (10.0,20.0,30.0,40.0,and 50.0µg per mL)versus transmittance.From the measured transmittance and the standard response line,determine the ascorbic acid concentration,C,in µg per mL,of the Assay Preparation.Calculate the quantity,in mg,of C6H8O6in the portion of liquids,tablets,or capsule contents taken by the appropriate formula:
For Liquids:
5C/Vin which Vis the volume,in mL,of liquid preparation taken to prepare the Assay Preparation.
For Tablets or Capsules:
12.5C.
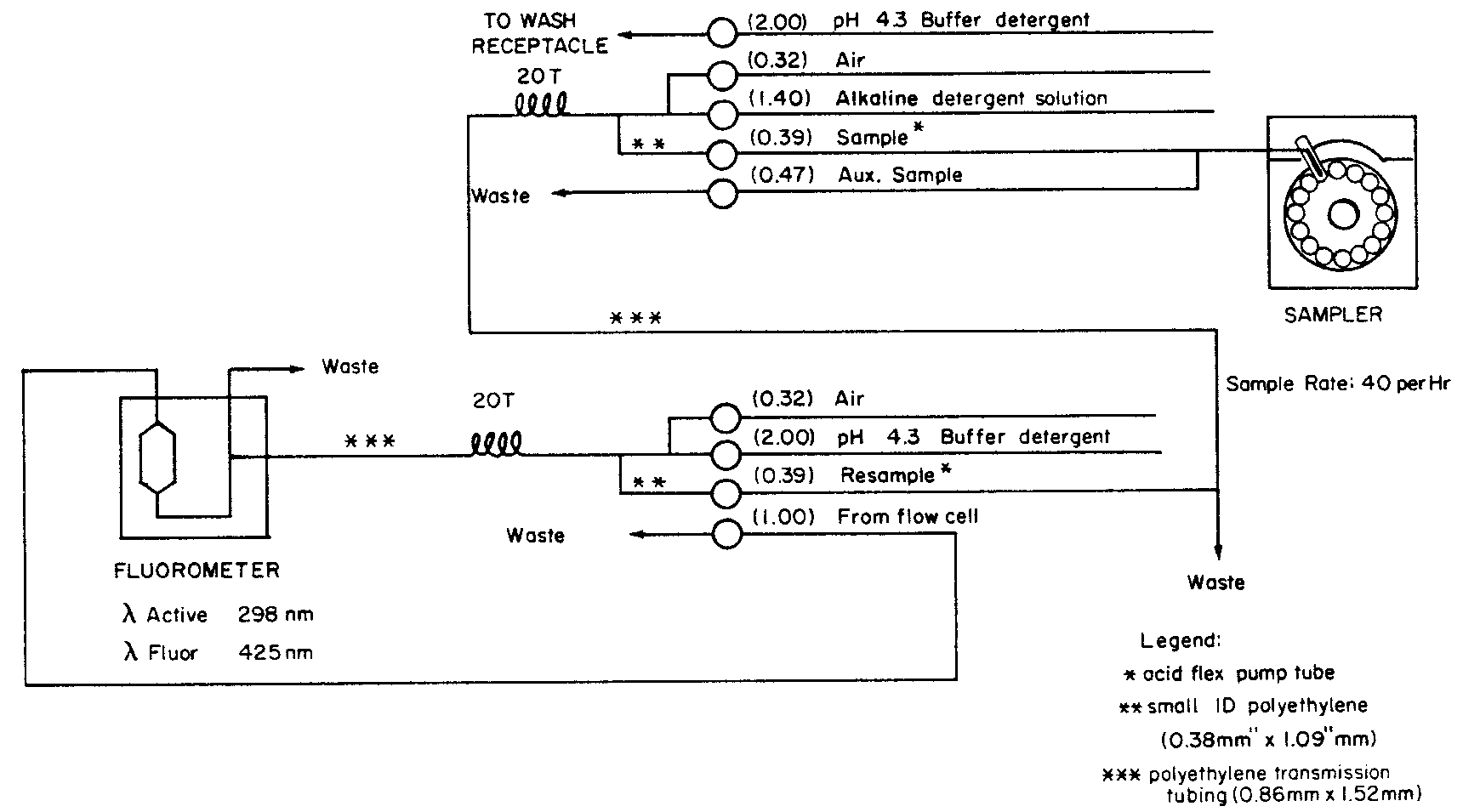
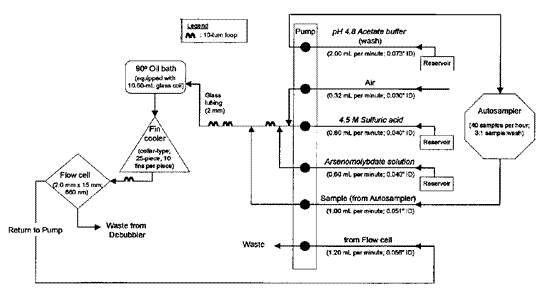
ASSAY FOR IODIDE
Apparatus—
Automatic analyzer consisting of (1)a liquid sampler,(2)a proportioning pump,(3)a heating bath,(4)a suitable colorimeter equipped with a 2.0-×50-mm flow cell and analysis capability at 420nm,(5)a means of recording colorimetric readings,and (6)a manifold consisting of the components illustrated in the accompanying pertinent diagram.
Reagents—
Acetic Acid Carrier Solution—
Transfer 3.0mLof glacial acetic acid to a 2000-mLvolumetric flask containing about 800mLof water.Add 2mLof polyoxyethylene (23)lauryl ether,and dilute with water to volume.
Surfactant Solution—
Prepare a 30%solution of polyoxyethylene (23)lauryl ether by melting 150g in a container on a steam bath and slowly adding approximately 250mLof water with continuous stirring.Cool,and dilute with water to make 500mL.
Arsenious Acid Solution—
Transfer 19.6g of arsenic trioxide and 14.0g of sodium hydroxide to a 2000-mLvolumetric flask.Add about 150mLof water,and dissolve with stirring.Dilute with water to a volume of about 800mL,and add 66mLof sulfuric acid.Cool to room temperature.Transfer 50.0g of sodium chloride to the solution,and mix to dissolve.Add 2mLof Surfactant Solution,dilute with water to volume,mix,and filter.
Ceric Ammonium Sulfate Solution—
Transfer 12.65g of ceric ammonium sulfate to a 1000-mLvolumetric flask.Add about 700mLof water followed by 100mLof sulfuric acid,swirling to mix.Heat to dissolve,and cool to room temperature.Add 1mLof Sulfactant Solution,dilute with water to volume,mix,and filter.
3%Acetic Acid Solution—
Transfer 30mLof glacial acetic acid to a 1000-mLvolumetric flask containing about 300mLof water.Dilute with water to volume,and mix.
Standard Preparations—
Standard Stock Solution—
Transfer an accurately weighed quantity of 1.3080g of potassium iodide,previously dried for 24hours at 105 ,to a 1000-mLvolumetric flask.Dilute with water to volume,and mix to obtain a solution having an iodide concentration of 1000µg per mL.
,to a 1000-mLvolumetric flask.Dilute with water to volume,and mix to obtain a solution having an iodide concentration of 1000µg per mL.
Intermediate Standard Solution—
Quantitatively dilute a suitable volume of Stock Standard Solutionwith water to obtain a solution having an iodide concentration of 1µg per mL.
Working Standard Preparations—
Transfer 2.0,4.0,6.0,8.0,and 10.0mLof Intermediate Standard Solutionto separate 100-mLvolumetric flasks.Add 5mLof 3%Acetic Acid Solution.Dilute the contents of each flask with water to volume,and mix to obtain Standard Preparations A,B,C,D,and Ehaving known iodide concentrations of about 0.02µg per mL,0.04µg per mL,0.06µg per mL,0.08µg per mL,and 0.1µg per mL,respectively.
Assay Preparation—
For Liquid Preparations—
Transfer an accurately measured volume of the liquid preparation,equivalent to 16µg of iodide,to a 200-mLvolumetric flask.Add 10mLof 3%Acetic Acid Solutionto dissolve,dilute with deionized water to volume,mix,and filter.Transfer 10.0mLof this solution to a 100-mLvolumetric flask,add 5.0mLof 3%Acetic Acid Solution,dilute with deionized water to volume,mix,and filter to obtain a solution having an iodide concentration of about 0.08µg per mL.
For Tablet Preparations—
Weigh and finely powder not fewer than 20Tablets.Transfer an accurately weighed quantity of the powder,equivalent to about 250µg of iodide,to a 250-mLvolumetric flask.Add 100mLof 1Nhydrochloric acid,and mix with the aid of sonication for 30minutes.Dilute with water to volume,mix,and filter.Transfer 8.0mLof the filtered solution to a 100-mLvolumetric flask,add 5mLof 3%Acetic Acid Solution,dilute with water to volume,and mix to obtain a solution having an iodide concentration of about 0.08µg per mL.
For Capsule Preparations—
Empty the contents,if necessary by cutting open with a sharp blade,of not fewer than 20Capsules into a suitable container,and mix thoroughly.Transfer a portion of the capsule contents,equivalent to about 250µg of iodide,to a 250-mLvolumetric flask and proceed as directed for Tablets above,beginning with “Add 100mLof .”
Procedure—
With the sample line pumping the Acetic Acid Carrier Solution,the other lines pumping their respective reagents,and the colorimeter equipped with 420-nm filters,standardize the system until a steady baseline has been established.Transfer portions of the Standard Preparationsand the Assay Preparationto the sampler cups,and place in the sampler.Start the sampler,and conduct determinations of each Standard Preparationand the Assay Preparationat the rate of 30per hour,using a ratio of about 1:4for sample and wash time.Derive a standard response line by plotting the respective Standard Preparationconcentration (0.02,0.04,0.06,0.08,and 0.10µg per mL)versus absorbance.[NOTE—This is an indirect absorbance relationship:the greater the iodide amount,the less the absorbance.]From the measured transmittance and the standard response line,determine the iodide concentration,C,in µg per mL,of the Assay Preparation.Calculate the quantity,in µg,of iodide in the portion of liquids,tablets,or capsules contents taken by the formula:
For Liquids:
2000C/Vin which Vis the volume,in mL,of the liquid preparation taken to prepare the Assay Preparation.
For Tabletsand Capsules:
3125C.
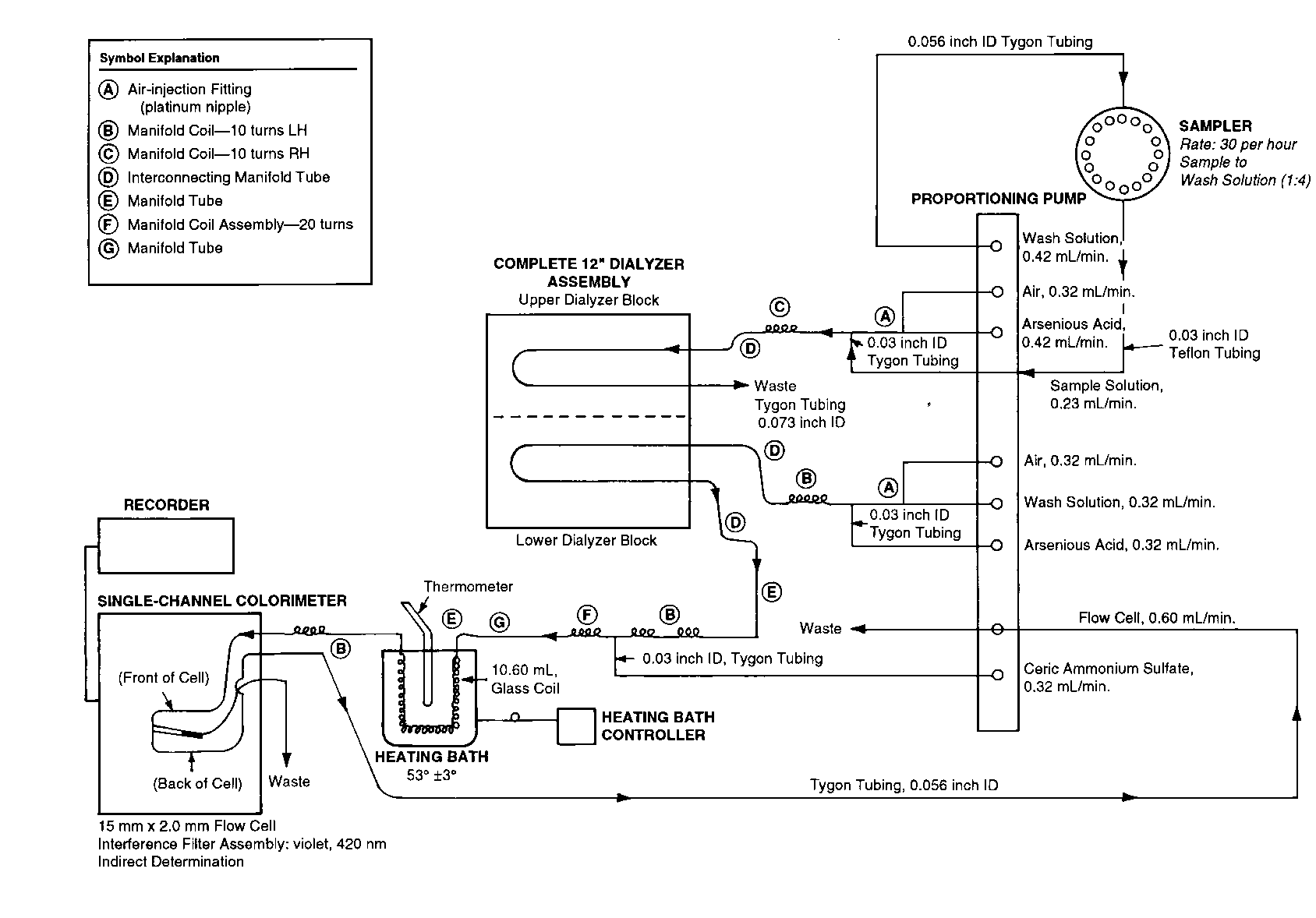
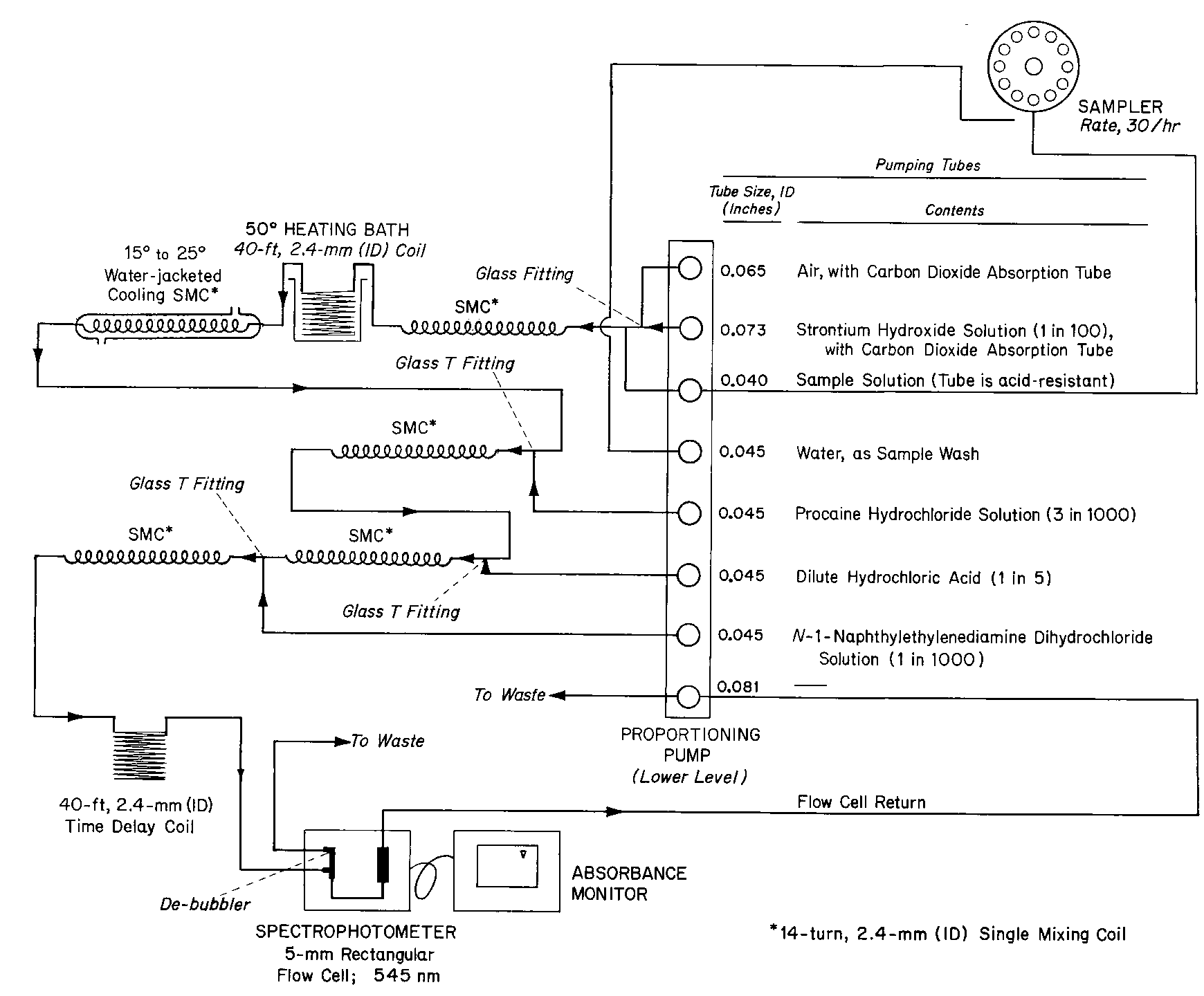
CONTENT UNIFORMITY OF NITROGLYCERIN TABLETS
This is not to be considered as the official method.It is detailed here for further illustration of descriptions of automated methods.
Apparatus—
Automatic analyzer consisting of (1)a liquid sampler,(2)a proportioning pump,(3)a heating bath,(4)a suitable spectrophotometer equipped with a 5-mm flow cell and analysis capability at 545nm,(5)a means of recording spectrophotometric readings,and (6)a manifold consisting of the components illustrated in the accompanying pertinent diagram.
Reagents—
1Percent Strontium Hydroxide Solution—
Dissolve 20.0g of strontium hydroxide [Sr(OH)2·8H2O]in 1800mLof carbon dioxide-free water,heating if necessary.Cool to room temperature,dilute with carbon dioxide-free water to 2000mL,and mix.Allow to stand overnight,and filter.Store the clear solution in tightly closed containers,protected from carbon dioxide.
0.3Percent Procaine Hydrochloride Solution—
Dissolve 3.0g of procaine hydrochloride in water to make 1000mL.
0.1Percent N-(1-Naphthyl)ethylenediamine Dihydrochloride Solution—
Dissolve 1.0g of N-(1-naphthyl)ethylenediamine dihydrochloride in water to make 1000mL.Prepare fresh each week.
Standard Preparation—
Dissolve an accurately weighed portion of 10%nitroglycerin-betalactose absorbate,previously standardized,in water,and dilute quantitatively and stepwise with water to obtain a solution having a known concentration of about 30µg per mL.
Test Preparation—
Dissolve 1Nitroglycerin Tablet in water to obtain a solution having a concentration of about 30µg of nitroglycerin per mL.
Procedure—
With the sample line pumping water,the other lines pumping their respective reagents,and the spectrophotometer set at 545nm,standardize the system by pumping until a steady absorbance baseline has been established.Transfer portions of the Standard Preparationand the Test Preparationto sampler cups,and place in the sampler.Start the sampler,and conduct determinations of the Standard Preparationand the Test Preparationat a rate of 30per hour,using a ratio of 1:1for sample and wash time.First,run two standards,discarding the first value,then continue the run using one standard after each five samples,recording the absorbance values.Calculate the quantity,in mg,of C3H5N3O9in the Tablet taken by the formula:
(T/D)C(AU/AS),
in which Tis the labeled quantity,in mg,of nitroglycerin in the Tablet;Dis the concentration,in µg per mL,of nitroglycerin in the solution from the Tablet,based on the labeled quantity per Tablet and the extent of dilution;Cis the concentration,in µg per mL,of nitroglycerin in the Standard Preparation;AUis the absorbance of the Test Preparation;and ASis the average of the absorbances of the two Standard Preparationsthat bracket the Test Preparation.
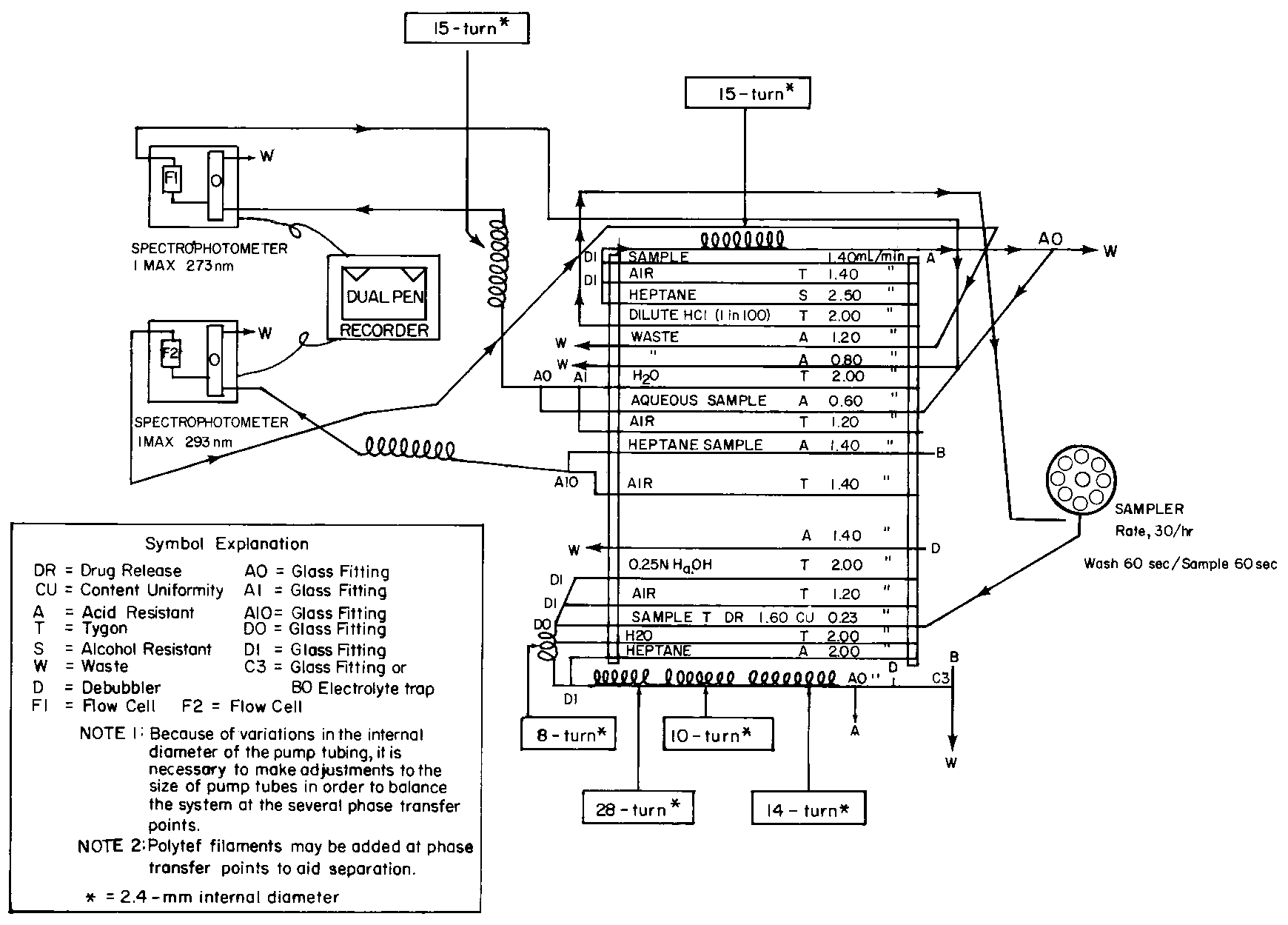
Diagram for Automated Drug Release and Content Uniformity Test for Propranolol Hydrochloride and Hydrochlorothiazide Extended-Release Capsules