 911
911 VISCOSITY
VISCOSITY
Viscosity is a property of liquids that is closely related to the resistance to flow. It is defined in terms of the force required to move one plane surface continuously past another under specified steady-state conditions when the space between is filled by the liquid in question. It is defined as the shear stress divided by the rate of shear strain. The basic unit is the poise; however, viscosities commonly encountered represent fractions of the poise, so that the centipoise (1 poise = 100 centipoises) proves to be the more convenient unit. The specifying of temperature is important because viscosity changes with temperature; in general, viscosity decreases as temperature is raised. While on the absolute scale viscosity is measured in poises or centipoises, for convenience the kinematic scale, in which the units are stokes and centistokes (1 stoke = 100 centistokes) commonly is used. To obtain the kinematic viscosity from the absolute viscosity, the latter is divided by the density of the liquid at the same temperature, i.e., kinematic viscosity = (absolute viscosity)/(density). The sizes of the units are such that viscosities in the ordinary ranges are conveniently expressed in centistokes. The approximate viscosity in centistokes at room temperature of ether is 0.2; of water, 1; of kerosene, 2.5; of mineral oil, 20 to 70; and of honey, 10,000.
Absolute viscosity can be measured directly if accurate dimensions of the measuring instruments are known, but it is more common practice to calibrate the instrument with a liquid of known viscosity and to determine the viscosity of the unknown fluid by comparison with that of the known.
Many substances, such as the gums employed in pharmacy, have variable viscosity, and most of them are less resistant to flow at higher flow rates. In such cases, a given set of conditions is selected for measurement, and the measurement obtained is considered to be an apparent viscosity. Since a change in the conditions of measurement would yield a different value for the apparent viscosity of such substances, the instrument dimensions and conditions for measurement must be closely adhered to by the operator.
Measurement of Viscosity—
The usual method for measurement of viscosity involves the determination of the time required for a given volume of liquid to flow through a capillary. Many capillary-tube viscosimeters have been devised, but Ostwald and Ubbelohde viscosimeters are among the most frequently used. Several types are described, with directions for their use, by the American Society for Testing and Materials (ASTM, D-445). The viscosity of oils is expressed on arbitrary scales that vary from one country to another, there being several corresponding instruments. The most widely used are the Redwood No. I and No. II, the Engler, the Saybolt Universal, and the Saybolt Furol. Each of these instruments uses arbitrary units that bear the name of the instrument. Standard temperatures are adopted as a matter of convenience with these instruments. For the Saybolt instruments, measurements usually are made at 100
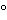
F and 210
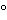
F; Redwood instruments may be used at several temperatures up to 250
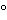
F; and values obtained on the Engler instrument usually are reported at 20
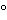
C and 50
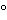
C. A particularly convenient and rapid type of instrument is a rotational viscosimeter, which utilizes a bob or spindle immersed in the test specimen and measures the resistance to movement of the rotating part. Different spindles are available for given viscosity ranges, and several rotational speeds generally are available. Other rotational instruments may have a stationary bob and a rotating cup. The Brookfield, Rotouisco, and Stormer viscosimeters are examples of rotating-bob instruments, and the MacMichael is an example of the rotating-cup instrument. Numerous other rotational instruments of advanced design with special devices for reading or recording, and with wide ranges of rotational speed, have been devised.
Where only a particular type of instrument is suitable, the individual monograph so indicates.
For measurement of viscosity or apparent viscosity, the temperature of the substance being measured must be accurately controlled, since small temperature changes may lead to marked changes in viscosity. For usual pharmaceutical purposes, the temperature should be held to within ±0.1
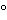
.
Procedure for Cellulose Derivatives—
Measurement of the viscosity of solutions of the high-viscosity types of methylcellulose is a special case, since they are too viscous for the commonly available viscosimeters. The Ubbelohde viscosimeter may be adapted (cf. ASTM, D-1347) to the measurement of the ranges of viscosity encountered in methylcellulose solutions.
Calibration of Capillary-Type Viscosimeters—
Determine the viscosimeter constant,
k, for each viscosimeter by the use of an oil of known viscosity.
*
Ostwald-Type Viscosimeter—
Fill the tube with the exact amount of oil (adjusted to 20.0 ± 0.1
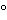
) as specified by the manufacturer. Adjust the meniscus of the column of liquid in the capillary tube to the level of the top graduation line with the aid of either pressure or suction. Open both the filling and capillary tubes in order to permit the liquid to flow into the reservoir against atmospheric pressure.
[NOTE—Failure to open either of these tubes will yield false values.
] Record the time, in seconds, for liquid to flow from the upper mark to the lower mark in the capillary tube.
Ubbelohde-Type Viscosimeter—
Place a quantity of the oil (adjusted to 20.0 ± 0.1
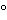
) in the filling tube, and transfer to the capillary tube by gentle suction, taking care to prevent bubble formation in the liquid by keeping the air vent tube closed. Adjust the meniscus of the column of liquid in the capillary tube to the level of the top graduation line. Open both the vent and capillary tubes in order to permit the liquid to flow into the reservoir against atmospheric pressure.
[NOTE—Failure to open the vent tube before releasing the capillary tube will yield false values.
] Record the time, in seconds, for the liquid to flow from the upper mark to the lower mark in the capillary tube.
Calculations—
Calculate the viscosimeter constant, k, from the equation:
k = v / d t,
in which
v is the known viscosity of the liquid in centipoises,
d is the specific gravity of the liquid tested at 20
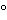
/20
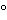
, and
t is the time in seconds for the liquid to pass from the upper mark to the lower mark.
If a viscosimeter is repaired, it must be recalibrated, since even minor repairs frequently cause significant changes in the value of its constant, k.
*
Oils of known viscosities may be obtained from the Cannon Instrument Co., Box 16, State College, PA 16801. For methylcellulose, choose an oil the viscosity of which is as close as possible to that of the type of methylcellulose to be determined.
Auxiliary Information—
Staff Liaison :
Catherine Sheehan, B.Sc., Scientist
Expert Committee : (EGC05) Excipient General Chapters
USP29–NF24 Page 2785
Phone Number : 1-301-816-8262

