Identification—
Prepare a solution of it containing about 12,000 Penicillin G Units per mL in a solvent mixture consisting of acetone, 0.1
M citric acid, and 0.1
M sodium citrate (2:1:1). Prepare a Standard solution of
USP Penicillin G Potassium RS containing about 12,000 Penicillin G Units per mL in the same solvent mixture (
Standard solution A). Prepare a Standard solution of
USP Procaine Hydrochloride RS containing about 5 mg per mL in the same solvent system (
Standard solution B). Apply separately 20 µL of each solution to a thin-layer chromatographic plate (see
Chromatography  621
621
) coated with a 0.25-mm layer of chromatographic silica gel mixture. Place the plate in a suitable chromatographic chamber, and develop the chromatogram in a solvent system consisting of a mixture of toluene, dioxane, and glacial acetic acid (90:25:4) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, mark the solvent front, and allow to air-dry. Examine the plate under short- and long-wavelength UV light, noting the positions of the spots. Spray the plate with
starch TS followed by dilute iodine TS (1 in 10). Penicillin G appears as a white spot on a purple background: the
RF value of the penicillin G spot obtained from the test solution corresponds to that obtained from
Standard solution A. Spray the location of the spots visualized with UV light with a 1 in 20 solution of
p-dimethylaminobenzaldehyde in methanol. Procaine appears as a bright yellow spot: the
RF value of the procaine spot obtained from the test solution corresponds to that obtained from
Standard solution B.
Content of Penicillin G and procaine—
Mobile phase—
Dissolve 14 g of monobasic potassium phosphate and 6.5 g of tetrabutylammonium hydroxide solution (4 in 10) in about 700 mL of water, adjust with 1 N potassium hydroxide to a pH of 7.0, dilute with water to 1000 mL, and mix. Mix 500 mL of this solution, 250 mL of acetonitrile, and 250 mL of water. Adjust with 1 N potassium hydroxide or dilute phosphoric acid (1 in 10) to a pH of 7.5 ± 0.05, filter through a membrane filter of 5 µm or finer porosity, and degas. Make adjustments if necessary (see
System Suitability under
Chromatography  621
621
).
Test preparation—
Transfer about 70 mg of Penicillin G Procaine, accurately weighed, to a 50-mL volumetric flask, add about 30 mL of Mobile phase, sonicate to dissolve, dilute with Mobile phase to volume, and mix.
Resolution solution—
Prepare a solution of penicillin V potassium in Mobile phase containing 2.4 mg per mL. Mix 1 volume of this solution and 3 volumes of Standard preparation.
Chromatographic system
(see
Chromatography  621
621
)—The liquid chromatograph is equipped with a 235-nm detector and a 4-mm × 30-cm column that contains 10-µm packing L1. The flow rate is about 1 mL per minute. Chromatograph the
Standard preparation, and record the peak responses as directed for
Procedure: the relative standard deviation for replicate injections is not more than 3.0%. Chromatograph about 10 µL of the
Resolution solution, and record the peak responses as directed for
Procedure: the resolution,
R, between penicillin G and penicillin V is not less than 2.0.
Procedure—
Separately inject equal volumes (about 10 µL) of the
Standard preparation and the
Test preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. The relative retention times are 1.0 for procaine and about 2.2 for penicillin G. Calculate the percentage of penicillin G (C
16H
18N
2O
4S) in the specimen under test by the formula:
50C(GS / WU)(rU / rS),
in which
C is the concentration, in mg per mL, of
USP Penicillin G Potassium RS in the
Standard preparation,
GS is the designated penicillin G content, in percentage, of
USP Penicillin G Potassium RS,
WU is the amount, in mg, of Penicillin G Procaine taken, and
rU and
rS are the responses of the penicillin G peaks obtained from the
Test preparation and the
Standard preparation, respectively: between 51.0% and 59.6% of C
16H
18N
2O
4S is found. Calculate the percentage of procaine (C
13H
20N
2O
2) in the specimen under test by the formula:
(236.32 / 272.78)(5000C / WU)(rU / rS),
in which 236.32 and 272.78 are the molecular weights of procaine and procaine hydrochloride, respectively,
C is the concentration, in mg per mL, of
USP Procaine Hydrochloride RS in the
Standard preparation,
WU is the amount, in mg, of Penicillin G Procaine taken, and
rU and
rS are the responses of the procaine peaks obtained from the
Test preparation and the
Standard preparation, respectively: between 37.5% and 43.0% is found.
Assay—
Assay preparation—
Prepare as directed for
Assay Preparation under
Iodometric Assay—Antibiotics  425
425
, except to dissolve about 100 mg of Penicillin G Procaine, accurately weighed, in 2.0 mL of methanol, and to dilute quantitatively with
Buffer No. 1 to obtain a solution containing about 2000 Penicillin G Units per mL.
Procedure—
Proceed as directed for
Procedure under
Iodometric Assay—Antibiotics  425
425
. Calculate the potency, in Penicillin G Units per mg, of the Penicillin G Procaine taken by the formula:
F(
B 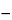 I
I) / (2
D),
in which
D is the concentration, in mg per mL, of the
Assay preparation, on the basis of the weight of Penicillin G Procaine taken and the extent of dilution, and the other terms are as defined therein.

