Add the following:
» Propofol Injectable Emulsion contains Propofol in a 10 percent (w/v) oil-in-water sterile emulsion. The aqueous component contains glycerol, a suitable antimicrobial agent, and Water for Injection. It contains not less than 90.0 percent and not more than 110.0 percent of the labeled amount of propofol (C12H18O). It contains a suitable emulsifying agent.
Packaging and storage—
Preserve under an inert atmosphere of nitrogen. Store at controlled room temperature. Do not freeze.
Labeling—
Label it to include the following: Shake well before use. Do not use if there is evidence of excessive creaming or aggregation, if large droplets are visible, or if there are other forms of phase separation indicating that the stability of the product has been compromised. Slight creaming, which should disappear after shaking, may be visible upon prolonged standing.
USP Reference standards  11
11 —
—
USP Endotoxin RS.
USP Propofol RS .
.
USP Propofol Related Compound A RS .
.
USP Propofol Related Compound B RS .
.
USP Endotoxin RS.
USP Propofol RS
 .
.
USP Propofol Related Compound A RS
 .
.
USP Propofol Related Compound B RS
 .
.
Identification—
A: Ultraviolet Absorption  197U
197U —
—
Spectral range:
200 to 450 nm.
Test solution—
Dilute a volume of Injectable Emulsion, equivalent to 10 mg propofol, with isopropyl alcohol to 100 mL.
Standard solution:
100 µg per mL.
Medium:
isopropyl alcohol.
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
Bacterial endotoxins  85
85 —
It contains not more than 0.33 USP Endotoxin Units per mg of propofol.
—
It contains not more than 0.33 USP Endotoxin Units per mg of propofol.
Sterility  71
71 :
meets the requirements.
:
meets the requirements.
pH  791
791 :
between 7.0 and 8.5.
:
between 7.0 and 8.5.
Globule size distribution in lipid injectable emulsions  729
729 :
meets the requirements.
:
meets the requirements.
Limit of free fatty acids—
[NOTE—In order to obtain a titer of sufficient sensitivity for the blank reagent, it is necessary to add stearic acid at a concentration of approximately 1.0 mMol per L.]
Blank titration—
Accurately weigh about 142.3 mg of stearic acid into a 500-mL volumetric flask. Dissolve in and dilute with dehydrated alcohol to volume to obtain the blank solution. Pipet 10 mL of the blank solution into a 100-mL beaker, and proceed as directed under Sample titration, beginning with “add 0.5 mL of 0.05 N hydrochloric acid”. Calculate the the blank correction, B, in mMol per L, by the formula:
(NVTB / VB) 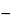 NSA
NSA
in which N is the concentration of the titrant, in mMol per L; VTB is the volume, in mL, of the titrant added between the first and second inflection points for the blank titration; VB is the volume, in mL, of the blank solution used for the analysis; and NSA is the concentration of stearic acid, in mMol per L.
Sample titration—
Transfer an accurate volume of Injectable Emulsion, equivalent to about 100 mg of propofol, to a 100-mL beaker, add 0.5 mL of 0.05 N hydrochloric acid, then dilute with a solution of alcohol and water (45:20) to 60 mL. Titrate with 0.05 M sodium hydroxide VS, determining the endpoint potentiometrically, using a suitable electrode. Calculate the free fatty acid content, in mMol per L, by the formula:
(NVT / VS) 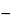 B
B
in which N is the concentration of the titrant, in mMol per L; VT is the volume, in mL, of the titrant added between the first and second inflection points; VS is the volume, in mL, of Injectable Emulsion used for the analysis; and B is the blank correction factor, in mMol per L, calculated as shown above: not more than 5 mMol per L is found.
Related compounds—
Mobile phase and Diluent—
Prepare as directed in the Assay.
Standard solution—
Dissolve accurately weighed quantities of USP Propofol RS, USP Propofol Related Compound A RS, and USP Propofol Related Compound B RS in Diluent, and dilute quantitatively, and stepwise if necessary, with Diluent to obtain a solution having known concentrations of about 0.8 mg per mL of propofol and 0.002 and 0.0008 mg per mL of propofol related compound A and propofol related compound B, respectively.
Test solution—
Use the Assay preparation, prepared as directed in the Assay.
Chromatographic system (see Chromatography  621
621 )—
Use the Chromatographic system as described in the Assay, except that detection is at 254 nm. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the relative retention times are about 0.8 for propofol related compound B, 1.0 for propofol, and 2.5 for propofol related compound A; the resolution, R, between propofol and propofol related compound B is not less than 2.5; the column efficiency is not less than 1000 theoretical plates based on the propofol peak; the tailing factor of the propofol peak is not more than 1.5; and the relative standard deviation for six replicate injections, based on the propofol peak, is not more than 2%.
)—
Use the Chromatographic system as described in the Assay, except that detection is at 254 nm. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the relative retention times are about 0.8 for propofol related compound B, 1.0 for propofol, and 2.5 for propofol related compound A; the resolution, R, between propofol and propofol related compound B is not less than 2.5; the column efficiency is not less than 1000 theoretical plates based on the propofol peak; the tailing factor of the propofol peak is not more than 1.5; and the relative standard deviation for six replicate injections, based on the propofol peak, is not more than 2%.
Procedure—
Separately inject equal volumes (about 20 µL) of the Test solution and the Standard solution into the chromatograph, record the chromatograms, and measure the peak responses. Calculate the content of propofol related compound A and of propofol related compound B, as a percentage of the label content of propofol, in the portion of Injectable Emulsion taken by the formula:
100(CS / CT) (ri / rS)
in which CS is the concentration, in mg per mL, of either propofol related compound A or propofol related compound B in the Standard solution; CT is the concentration, in mg per mL, of propofol in the Test solution based on the label claim; ri is the individual peak response of either propofol related compound A or propofol related compound B in the Test solution; and rS is the individual peak response of either propofol related compound A or propofol related compound B in the Standard solution: not more than 0.5% of propofol related compound A is found; and not more than 0.5% of propofol related compound B is found.
Other requirements—
It meets the requirements under Injections  1
1 .
.
Assay—
Mobile phase—
Prepare a filtered and degassed mixture of water and tetrahydrofuran (60:40). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Diluent—
Dilute 68 mL of water with isopropyl alcohol to 1 L, and mix.
Standard preparation—
Dissolve an accurately weighed quantity of USP Propofol RS in Diluent, and dilute quantitatively, and stepwise if necessary, with Diluent to obtain a solution having a known concentration of about 0.8 mg per mL of propofol.
Assay preparation—
Transfer a volume of well-shaken Injectable Emulsion, equivalent to about 40 mg of propofol, to a 50-mL volumetric flask, dissolve in and dilute with isopropyl alcohol to volume, and mix. The solution contains about 0.8 mg of propofol per mL.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 275-nm detector and a 5-mm × 10-cm column that contains 5-µm packing L1. The flow rate is about 2 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 1000 theoretical plates; the tailing factor of the propofol peak is not more than 1.5; and the relative standard deviation for replicate injections is not more than 2%.
)—
The liquid chromatograph is equipped with a 275-nm detector and a 5-mm × 10-cm column that contains 5-µm packing L1. The flow rate is about 2 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 1000 theoretical plates; the tailing factor of the propofol peak is not more than 1.5; and the relative standard deviation for replicate injections is not more than 2%.
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major propofol peaks. Calculate the quantity, in percent of label claim, of propofol (C12H18O) in the portion of Injectable Emulsion taken by the formula:
 USP32
USP32
100(CS / CU)(rU / rS)
in which CS is the concentration, in mg per mL, of propofol in the Standard preparation; CU is the concentration, in mg per mL, of propofol in the Assay preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Daniel K. Bempong, Ph.D.
Senior Scientist 1-301-816-8143 |
(MDPS05) Monograph Development-Pulmonary and Steroids |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
|
| Radhakrishna S Tirumalai, Ph.D.
Senior Scientist 1-301-816-8339 |
(MSA05) Microbiology and Sterility Assurance | |
| Radhakrishna S Tirumalai, Ph.D.
Senior Scientist 1-301-816-8339 |
(MSA05) Microbiology and Sterility Assurance |
USP32–NF27 Page 3413
Pharmacopeial Forum: Volume No. 33(6) Page 1208
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.